|
On the other hand, when an electron jumps from a higher energy level to a lower energy level, the hydrogen atom emits a photon. When a photon is absorbed by a hydrogen atom, the absorbed energy cause excitation of an electron from a low energy level to a high energy level. 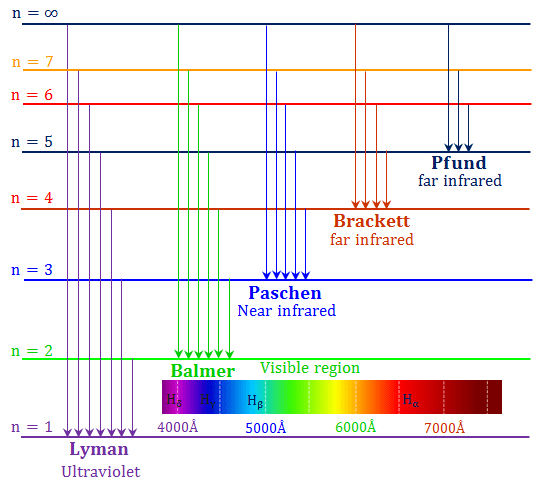
ν = wave number of the emitted radiation.Rydberg gave an expression to describe the wave number of emitted spectra The spectrum of a hydrogen atom is important to understand that energy levels of electrons are quantized. These frequencies produce a spectrum of a series of lines. When an electric discharge is passed through hydrogen gas (H 2), the molecules excite to form hydrogen atoms and emit light (radiation) of the discrete frequencies. It was all a follow-up of Bohr’s atomic model and through his explanation, scientists were able to develop an atomic theory with the least limitations. 
In 1890, Swedish spectroscopist Johannes Rydberg described the spectra of hydrogen atoms. This type of spectra would be either emission or absorption spectrum. These wavelengths are very sharp lines with well-defined boundaries. The line spectrum shows only a certain wavelength within a given range. Thus the spectrum in most media consists of a series of sharp lines with widths of 10 -1 – 10 -2 Å (10 -2 – 10 -3 nm). The individual particles in the gas medium behave independently of one another. Line spectrum occurs when wavelengths of radiation emitted or absorbed by a substance are well separated. It means that this spectrum has no visible boundaries or gaps between a range of frequencies or wavelengths. Through this, the VIBGYOR (violet, indigo, blue, green, yellow, orange, red) diversity sequence came to be known.Ī continuous spectrum has no line separation between two different colors yet they form a gradient. He separated sunlight into its component colors by using prisms and showed that sunlight consists of a continuous array of colors. In 1672, Isaac Newton was the first to discover that sunlight is made up of multiple frequencies or wavelengths. This is because the absorbed energy is the same as radiant energy. For a given atom, the absorbed lines in absorption spectra and emitted lines in emission spectra are in the same place. The emission spectrum is opposite to the absorption spectrum. The emission or bright-line spectrum of chemical species comprises a range of frequencies of electromagnetic radiation emitted due to the transition of an electron from its excited (high-energy)state to the ground (low-energy) state. The analysis of that spectra gives information about the identity and concentration of a specific substance. The absorption spectrum is plotted with absorbance against wavelength or frequency. It means that all atoms always absorb the same characteristic wavelength from given electromagnetic radiation. Every atom has its unique absorption and emission spectrum range because the energy difference is discrete and constant. 
The absorbed wavelengths leave dark spaces in the continuous spectrum. When electromagnetic radiation is passed through chemical species, certain wavelengths are absorbed. In the absorption spectrum, the electromagnetic radiation of certain frequencies is transmitted through an analyte with dark fringes or bands when the ground-state electrons absorb energy to get to higher levels or excited. The information about the analyte is obtained from the amount of EMR absorbed as a result of excitation or measuring the electromagnetic radiation emitted when the analyte turns back to the ground state from the excited state. When EMR of different energies interact with that analyte, some of the energy is absorbed and the chemical species undergo a transition to a higher (excited) state. These records are generally referred to as spectra.ĭifferent types of spectrometry and spectroscopy techniques are listed as:īefore the interaction of electromagnetic radiations, the analyte is in the ground state. Optical instruments called spectrometers reveal a series of specific wavelengths or frequencies in which the light energy is absorbed and emitted. When light of a certain wavelength passes through an object, some of it can be absorbed and the rest can be emitted at a different wavelength from its original one. The interaction between electromagnetic energy and matter is studied in the field of spectroscopy. The record of the intensity of light (radiations) transmitted or scattered by an atom or a molecule as a fraction of its frequency, wavelength, or wavenumber is called its spectrum (From Latin words ‘ spectre‘ and ‘ apparition‘ meaning ‘appearance’).
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
